
Working with CRM to move Malaysia forward
The Malaysian government has committed funding to improve the healthcare systems in order to further support clinical research. Clinical Research Malaysia (CRM) is a non-profit organisation &# […]
了解更多
The Malaysian government has committed funding to improve the healthcare systems in order to further support clinical research. Clinical Research Malaysia (CRM) is a non-profit organisation &# […]
了解更多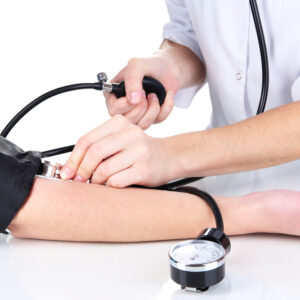
Globally, there are millions of people that could benefit from an intensive lowering of blood pressure (BP). An individual with lower BP decreases their chances of suffering from any cardiovascular events such as: stroke, heart disease or heart attacks. A new study shows that lowering blood pressure to less than 140 mm Hg, the current […]
了解更多
Over the last 10 years, South Korea has increasingly become a global clinical trial (CT) hub and has established itself as one of the leading CT destinations in the Asia region. In 2015, South […]
了解更多
New initiatives from the country’s regulator aim to smooth the registration of trials and ensure prospective participants fully understand what will be involved through mandatory AV recording of the consent process. Clinical trial applications in India are taking a step forward, with the approval of a Central Drugs Standard Control Organisation (CDSCO) IT-enabled system from […]
了解更多
Smartphones are changing our lives every day. Almost every industry is affected by the mobile revolution in one way or another. At 乔治临床( George Clinical ), we are in the business of clinical t […]
了解更多
When it comes to jobs in healthcare, working in the field of clinical research can be exciting and rewarding. There are many different opportunities available in the healthcare job field. If y […]
了解更多
Do you ever wonder how prescription medications make their way to your local pharmacy? Clinical trials and health research play a vital role in maintaining the world population’s health on a r […]
了解更多
Effective 2nd August 2016, the Central Drugs Standard Control Organization (CDSCO) India released new circulars in relation to the conduct of clinical trials. These new circulars have modified […]
了解更多
EDC has become the data management platform of choice around the world, offering advantages such as increasing the quality and timely collection of data. Medrio, an EDC company that offers eClinical software solutions, has quoted that 70% of clinical trials are using EDC as a forms of collecting data in the United States. In China, […]
了解更多